 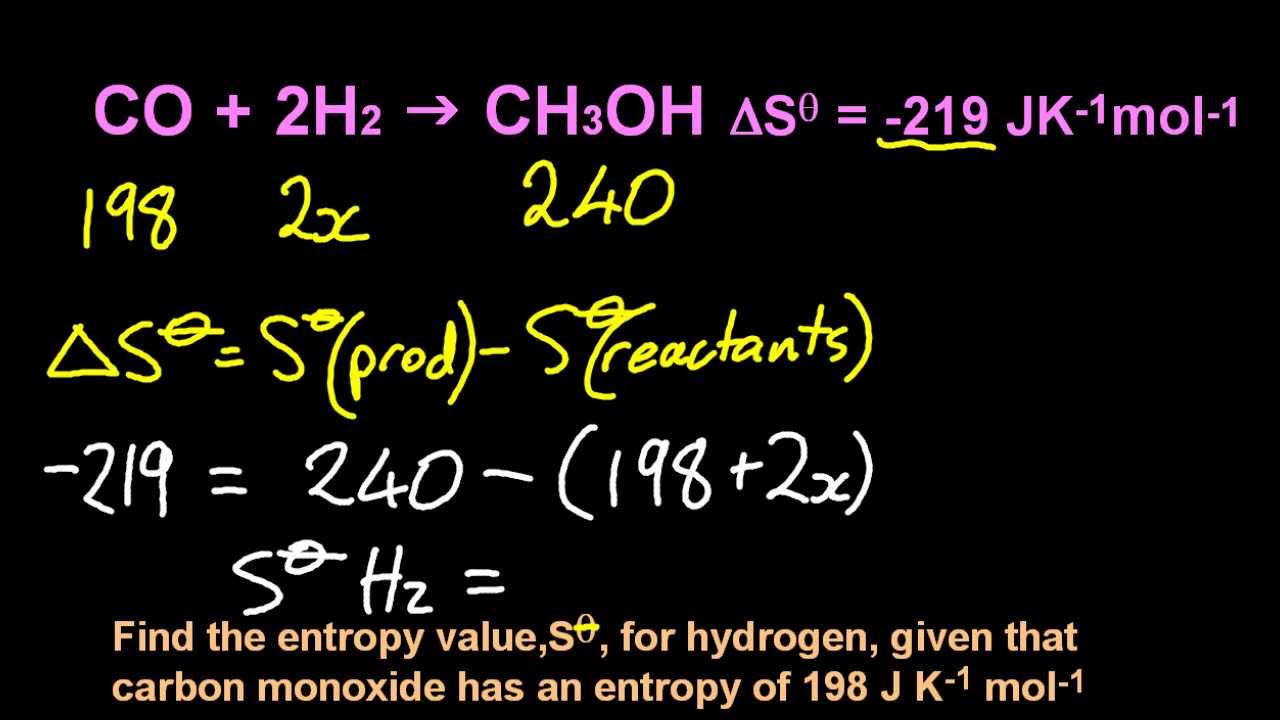
After the reaction, the two are bonded together and can't float around freely from one another. Solve any question of Chemical Thermodynamics with:. In other words the N 2( g) used to float around independently of the H 2 gas molecules. Entropy: A measure of the unavailable energy in a closed thermodynamic system that is also usually considered to be a measure of the systems disorder. This is expected because we are decreasing the number of gas molecules. It would appear that the process results in a decrease in entropy - i.e. \įrom the balanced equation we can write the equation for ΔS 0 (the change in the standard molar entropy for the reaction): As with other calculations related to balanced equations, the coefficients of each component must be taken into account in the entropy calculation (the n, and m, terms below are there to indicate that the coefficients must be accounted for): The entropy change in a chemical reaction is given by the sum of the entropies of the products minus the sum of the entropies of the reactants. Unlike enthalpies of formation, standard molar entropies of elements are not 0.When comparing standard molar entropies for a substance that is either a solid, liquid or gas at 298 K and 1 atm pressure, the gas will have more entropy than the liquid, and the liquid will have more entropy than the solid. 
A table of standard molar entropies at 0K would be pretty useless because it would be 0 for every substance (duh!) Standard molar entropy values are listed for a variety of substances in Table T2. the entropy of a pure substance at 298 K and 1 atm pressure).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
